Hair Growth + scalp health
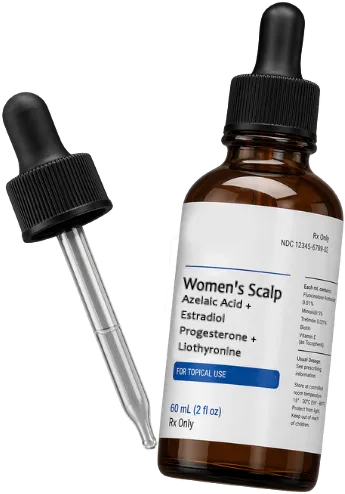
Women's Scalp Solution
4-in-1 hormonal scalp formula addressing the multifactorial biology of female hair loss in a single nightly application.
No Insurance Required
Ships From USA Pharmacy
Free Medical Consultation
No Subscription Required
Licensed Provider
Licensed Pharmacy
HIPAA Compliant
LegitScript Certified
Overview
Azelaic Acid + Estradiol + Progesterone
Liothyronine
Topical 5-alpha reductase inhibitor + estrogen receptor agonist + androgen receptor competitor + thyroid hormone receptor agonist. Targets all four primary hormonal axes of female hair loss — androgens, estrogen, progesterone, and thyroid — directly at the follicular level.
Mechanism of Action
Azelaic acid inhibits 5-alpha reductase to reduce intrafollicular DHT and lowers perifollicular oxidative and inflammatory burden. Estradiol activates estrogen receptors in the outer root sheath and dermal papilla to extend anagen and oppose androgenic follicular signaling. Progesterone competes at androgen receptors in the follicular unit to further suppress DHT-mediated miniaturization without systemic antiandrogen exposure. Liothyronine activates thyroid hormone receptors in dermal papilla cells, supporting metabolic activity, protein synthesis, and anagen maintenance at the follicular level.
Topical Scalp Solution
Applied once nightly to clean, dry affected scalp areas. Massaged thoroughly into thinning regions, typically the crown and midline part.
Information presented here is for educational purposes only and not intended to replace or substitute guidance from a healthcare provider. Compounded medications are not FDA-approved nor evaluated by the FDA for safety, efficacy, or quality.
Benefits
Hormonal Axis Coverage
Targets all four primary biological axes of female hair loss simultaneously: androgenic DHT excess, estrogen loss, progesterone depletion, and insufficient thyroid hormone signaling at the follicular level.
Scalp Health
Azelaic acid's anti-inflammatory and antioxidant activity creates a healthier perifollicular environment for the hormonal restoration delivered by the other three actives.
Shedding Reversal
Addresses hormonally driven hair thinning at its root, reducing the psychological burden of progressive hair loss in perimenopausal and postmenopausal women.
DHT Suppression
Dual anti-androgenic coverage from azelaic acid (enzyme inhibition) and progesterone (receptor-level competition) provides comprehensive DHT suppression without systemic HRT-level hormonal exposure.
Protocol Flexibility
Appropriate as a standalone hormonal scalp treatment or as a targeted adjunct in women already on systemic HRT who need additional local follicular hormonal support.
Individual results may vary. Benefits described are based on clinical and pharmacological evidence and do not constitute a guarantee of treatment outcomes. All treatment requires evaluation and approval by a licensed provider.
Simple. Medical. Personalized.

Medical intake
Answer a few online questions about your health history, lifestyle, & goals. No clinic visits required.
Provider evaluation
A licensed medical provider reviews your intake to determine the safest, personalized treatment.


Personalized plan
Custom treatment plan built around your unique health needs, goals, medical history, & biology.

Ongoing support
Stay connected with your care team for follow-ups, adjustments, & expert answers 100% online.
What Patients Are Experiencing

"My hair started thinning significantly after menopause. Six months on this formula and my ponytail is noticeably fuller. First thing that has actually worked."
— Barbara N. *

"My provider explained the thyroid connection to hair loss. Once we addressed that axis topically alongside the hormonal support, shedding dropped dramatically."
— Diane K. *

"Postpartum hair loss hit me hard. This addressed multiple factors my OB said could be contributing. Density improved and shedding normalized by month 4."
— Rachel A. *
*Individual experiences may vary. Not all patients will have the same outcomes.
FAQs
What is Women Scalp Solution?
Women Scalp Solution is a prescription therapy used for women's scalp and hair support.
Who should use Women Scalp Solution?
Patients seeking support for women's scalp and hair support may qualify after evaluation by a licensed provider.
Does Women's Scalp Solution require a prescription?
Yes. Women's Scalp Solution is a compounded prescription topical preparation requiring a valid prescription from a licensed healthcare provider. A telehealth consultation, hair loss evaluation, and hormone and health history review are required before dispensing to identify the cause of hair loss, confirm the appropriate active combination, and establish the treatment protocol.
What are the possible side effects?
Estradiol and progesterone: mild scalp irritation is most common. Rare systemic effects including breast tenderness, fluid retention, or mood changes may occur with significant absorption. Liothyronine: palpitations, tachycardia, or anxiety are possible if significant systemic absorption occurs, particularly in thyroid-sensitive patients. Azelaic acid: mild scalp tingling, warmth, or dryness in the first one to two weeks. Transient increased shedding in weeks 4-8 as follicles re-enter anagen is expected and temporary.
Who Should Not Use Women's Scalp Solution?
Women who are pregnant or may become pregnant should discuss all hair restoration actives with their provider, as certain agents (finasteride, spironolactone, and others) carry fetal risk. Patients with active scalp infections, significant inflammatory scalp conditions, or hypersensitivity to any prescribed active should not use this formulation until these conditions are evaluated. Those with a history of hormone-sensitive malignancy should have any hormonally active components reviewed with their oncologist before starting.
Are there any drug interactions?
Patients on systemic HRT (oral or transdermal estradiol, progesterone) require coordinated provider management. Patients on any systemic thyroid medication (levothyroxine, desiccated thyroid, or liothyronine) require thyroid function monitoring and potential dose adjustment. Antithyroid medications (methimazole, propylthiouracil) may be partially counteracted. Beta-blockers and cardiovascular medications warrant disclosure due to the thyroid component.
Is Women's Scalp Solution safe if I have a pre-existing medical condition?
Patients with thyroid disease, cardiovascular conditions, hormone-sensitive malignancy history, undiagnosed uterine bleeding, or liver impairment require individualized provider evaluation before starting. Lab monitoring including thyroid function and hormonal levels is recommended during treatment.
Contact Us
Start Your Journey
Ready to Address Your
Hair Loss at Every Level?
Physician-prescribed. Delivered to your door. No clinic visits required.
Licensed Provider
Licensed Pharmacy
HIPAA Compliant
LegitScript Certified

© 2026 TestosteroneShots.com, PC
text (323)-283-9219
638 1/2 N. Robertson Blvd, West Hollywood, CA 90069
Legal
The information and clinical services described on this website are for educational and informational purposes only and are not intended to diagnose, treat, cure, or prevent any disease. All treatments require evaluation and approval by a licensed healthcare provider through a telemedicine consultation. Treatment approval is not guaranteed. Individual results may vary, and treatments may carry risks and side effects. Certain compounded medications, wellness treatments, or off-label uses may not be evaluated by the U.S. Food and Drug Administration (FDA) for safety, effectiveness, or quality unless explicitly stated. Testosterone Shots provides telehealth consultations, clinical review, care coordination, lab-related services, membership services, and ongoing treatment management. Testosterone Shots is not a pharmacy and does not manufacture, compound, dispense, sell, handle, warehouse, ship, or collect payment for prescription medication. Testosterone Shots collects payment only for consultations, clinical review, care coordination, lab-related services, membership services, and other non-pharmacy services. If treatment is approved, your prescription may be sent to a licensed pharmacy partner. The pharmacy contacts you directly to complete medication payment and shipping. Services are provided by licensed physicians, nurse practitioners, and/or other licensed healthcare providers in states where they are authorized to practice. This service is not intended for medical emergencies. If you are experiencing an emergency, call 911 or seek immediate medical care. We take reasonable measures to protect personal health information in accordance with applicable privacy laws, including HIPAA. By using this website or its services, you agree to our Terms of Service, Privacy Policy, Notice of Privacy Practices, Telehealth Consent, and Important Safety Information. You must be 18 years of age or older to use this service.

