

Consult Licensed Providers. Start Treatment From home.
Testosterone
Optimization with Testosterone
Cypionate & Pregnyl (hCG)

Anti-Aging
Vitamins & Amino Acids
Boost Cellular Longevity

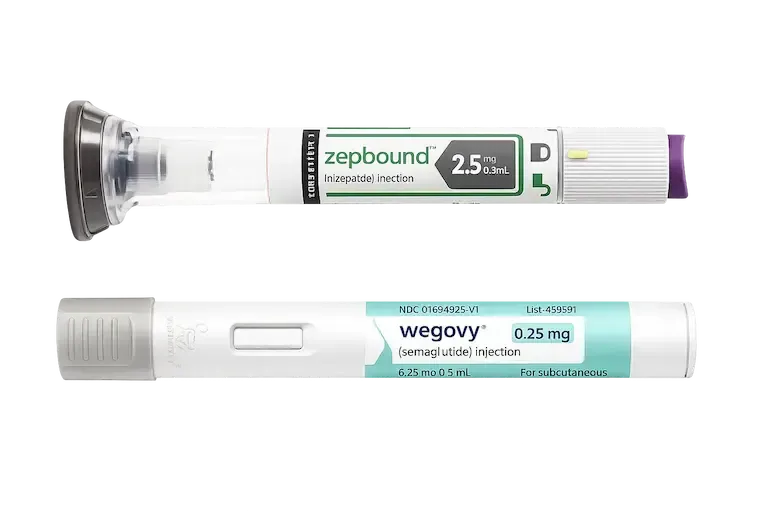
Weight Loss
Wegovy® & Semaglutide
Zepbound® & Tirzepatide
Brain Focus
Unlock Sharper Focus, Memory, & Mental Clarity
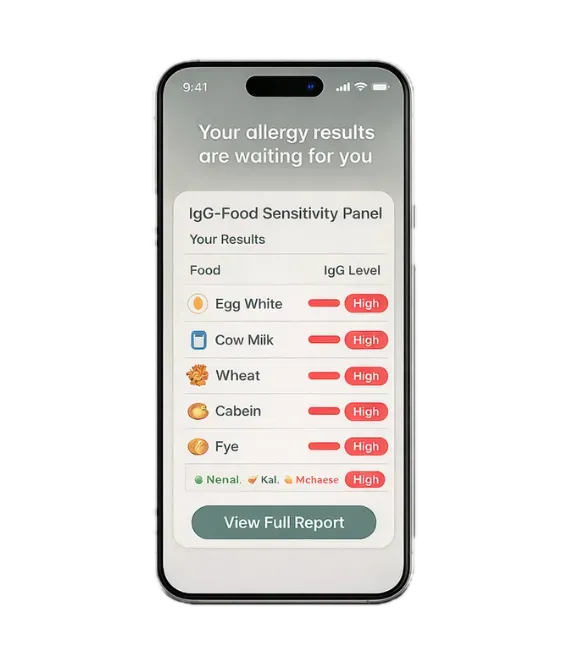
Food & Gut Allergies
Discover What's Irritating
Your Gut with an At-Home Test
Biohacking
Advanced Biomarker &
Genetic Labs

Hair Restoration
Repair Scalp Damage
Vascularize Hair Follicles

Sleep
Medical Sleep Support for
Deeper, More Consistent Rest

Skincare
Target Wrinkles, Rosacea,
Eczema, & Acne Scars

Blood Work
Check Hormones, Allergies, Biological Age, & Organ Health

Sexual Health
Sildenafil, Tadalafil, Oxytocin,
PT-141. Fast-Acting

Supplements
20% off Top Clinical Brands:
Thorne® Pure® Metagenics®

Interested in Prescription Peptides?
Peptides are short chains of amino acids that signal your body to repair, recover, and perform. Learn the science behind them.

Interested in Prescription Peptides?
Peptides are short chains of amino acids that signal your body to repair, recover, and perform. Learn the science behind them.
Explore by Rx Treatment
How It Works
Choose a treatment
Browse a curated selection of medically guided treatments designed for real needs, not trends. Each option is clearly explained so you can make an informed choice without medical jargon or confusion.
Complete a secure medical intake
Fill out a confidential health questionnaire that helps our providers understand your medical history,
goals, and current condition. Your information is encrypted and reviewed with strict privacy standards.

Provider review & approval
A licensed medical provider reviews your intake to ensure the treatment is personalized to your goals. They may request additional details or lab work before approval.

Prescription Sent to Pharmacy
Once approved, your prescription is sent to a licensed pharmacy for fulfillment. The pharmacy will prepare and ship your medication discreetly to you, fitting seamlessly into your routine.
Private. Simple. Delivered.
100% Online Medical
Receive professional medical care entirely online, from consultation to prescription management. No waiting rooms, no appointments to commute to - everything happens on your schedule.
Pharmacy Shipping

Your privacy comes first. All medications are shipped in plain, unbranded packaging with no indication of contents, ensuring a confidential and stress-free delivery experience.
Ongoing Support

Care doesn’t stop once you receive your medication. Stay connected with licensed providers for follow-ups, dosage adjustments, refills, and questions through your secure patient portal.
Trust & Compliance
HIPAA-Compliant Platform
We use secure, HIPAA-compliant systems to protect your personal and medical data at every step, from intake forms to ongoing care. Your information is never shared without authorization.
Licensed Medical Providers
All treatments are reviewed and approved by licensed medical professionals who follow
established clinical guidelines and regulatory requirements.
Partner Licensed Pharmacies
Prescriptions are fulfilled by licensed 503A or 503B compounding pharmacies that meet strict quality, safety, and compliance standards.
Proof, Not Promises
High Standards
Higher Ratings

Our Experts

Dr. Gideon Kwok, DO
Medical Director

Ahmed Mahdi, DNP
Practitioner
Our Partners







FAQs
Are you a pharmacy?
Testosterone Shots is a telehealth platform, not a pharmacy. We provide online consultations, clinical review, and care coordination. If treatment is approved, your prescription is sent to a licensed pharmacy partner, who handles medication payment, fulfillment, and shipping directly to you.
Do I need blood work?
Our medical providers recommend a baseline lab panel for a personalized treatment plan based on your biomarkers. This helps us understand your unique health profile, prescribe the right treatment at the right dose, and monitor your progress over time. We offer 16 lab panels starting at $99 through Quest Diagnostics, with results in 3-5 business days.
How does the process work?
Complete a secure medical intake form and consult with a licensed provider through telehealth. If treatment is approved, your prescription may be sent to a licensed pharmacy partner. The pharmacy contacts you directly to verify shipping information, verify billing information, and collect payment for the medication. The pharmacy then dispenses and ships the medication directly to you, where permitted by law.
How long does it take to get my medication?
After provider approval, timing depends on the pharmacy. Most patients receive pharmacy-shipped medications within 5–7 business days after the pharmacy completes payment and shipping confirmation.
When will I start feeling results?
Results depend on the treatment and individual health profile. Every patient's experience is different and results may vary. Your provider will discuss realistic expectations during your consultation and adjust your treatment plan based on your progress and lab work.
Who will I see for my consultation?
All consultations are conducted by licensed medical providers, including DNPs, MDs, or DOs, who specialize in each treatment area.
Do you accept insurance?
We do not accept insurance at this time. Patients may be able to use insurance at Quest Diagnostics or Labcorp to help cover the cost of blood work.
What if I'm not approved for treatment?
Our goal is to help you become healthy. Under the guidance of our medical providers, we personalize a treatment plan tailored to your goals, biomarkers, and DNA. If a specific treatment isn't right for you, a provider will recommend alternative options to help you reach your health goals.
What is your refund policy?
Consultation, clinical review, care coordination, membership, and lab-related fees may be subject to the refund terms listed in our Terms of Service. Testosterone Shots does not collect payment for prescription medication and does not dispense, ship, or accept returns of prescription medication. Medication billing, payment, dispensing, shipping, returns, and refund issues are handled directly by the dispensing pharmacy, subject to pharmacy policy and applicable law.
Contact Us
Start Your Journey
Finally feel like
yourself again
Our doctors listen first. No clinic visits required.
Licensed Provider
Licensed Pharmacy
HIPAA Compliant
LegitScript Certified

© 2026 TestosteroneShots.com, PC
text (323)-283-9219
638 1/2 N. Robertson Blvd, West Hollywood, CA 90069
Legal
The information and clinical services described on this website are for educational and informational purposes only and are not intended to diagnose, treat, cure, or prevent any disease. All treatments require evaluation and approval by a licensed healthcare provider through a telemedicine consultation. Treatment approval is not guaranteed. Individual results may vary, and treatments may carry risks and side effects. Certain compounded medications, wellness treatments, or off-label uses may not be evaluated by the U.S. Food and Drug Administration (FDA) for safety, effectiveness, or quality unless explicitly stated. Testosterone Shots provides telehealth consultations, clinical review, care coordination, lab-related services, membership services, and ongoing treatment management. Testosterone Shots is not a pharmacy and does not manufacture, compound, dispense, sell, handle, warehouse, ship, or collect payment for prescription medication. Testosterone Shots collects payment only for consultations, clinical review, care coordination, lab-related services, membership services, and other non-pharmacy services. If treatment is approved, your prescription may be sent to a licensed pharmacy partner. The pharmacy contacts you directly to complete medication payment and shipping. Services are provided by licensed physicians, nurse practitioners, and/or other licensed healthcare providers in states where they are authorized to practice. This service is not intended for medical emergencies. If you are experiencing an emergency, call 911 or seek immediate medical care. We take reasonable measures to protect personal health information in accordance with applicable privacy laws, including HIPAA. By using this website or its services, you agree to our Terms of Service, Privacy Policy, Notice of Privacy Practices, Telehealth Consent, and Important Safety Information. You must be 18 years of age or older to use this service.