

Consult licensed providers, get prescribed treatments, & receive medications at home.
Testosterone
Optimization with Testosterone Cypionate and Pregnyl (HCG)

Anti-Aging
Vitamins, amino acids designed to boost cellular performance

Weight Loss
Wegovy® & Semaglutide
Zepbound® & Tirzepatide
Brain Focus
Unlock sharper focus, memory, and mental edge
Food & Gut Allergies
Discover what's irritating your gut - test from home
Biohacking
Advanced biomarker labs, organ health and DNA

Hair Restoration
Repair scalp damage and vascularize hair follicles

Sleep
Medical sleep support for deeper, more consistent rest

Skincare
Target wrinkles, rosacea, eczema, and acne scars

Blood Work
Check hormone levels, biomarkers, biological age

Gooning ~ Sex
Sildenafil, Tadalafil, Oxytocin, PT-141. Fast-Acting

Supplements
20% off top clinical brands: Thorne®, Pure®, Metagenics®

Peptides
Heal faster, Recover stronger, Age slower
Explore by Rx Treatment
immune

Tri-Immune Boost
Vitamin C, glutathione & zinc — three immune powerhouses in one injectable shot.
Supercharge immune defense fast
Detox, skin health & cellular repair
Recover quicker from illness or stress
$150
collagen

Amino Blend
Arginine, citrulline, lysine & proline — nitric oxide precursors and collagen builders.
Maximize blood flow and pump
Strengthen tendons, skin, and vessels
Faster recovery between workouts
$150
hair skin nails

Vitamin B-Complex
All 8 essential B vitamins — the cofactors that drive energy metabolism and nerve function.
Eliminate brain fog and fatigue
Support mood and stress resilience
Fuel every cell in your body
$160
testosterone boost

Pregnyl (HCG)
Human chorionic gonadotropin — mimics luteinizing hormone.
Naturally produces more testosterone
Prevent testicular atrophy on TRT
Increases sperm count
$350
testosterone boost
T-Booster
4 in 1: Enclomiphene, DHEA, Pregnenolone & Zinc to raise testosterone naturally.
Boost energy & mental drive
No injections, oral capsule
Increase sperm and libido
$120
trt

Testosterone & HCG
Gold standard for testosterone replacement therapy, including HCG.
Regain energy levels & self-esteem
Pregnyl (HCG)
Testosterone Cypionate
$200/month
Youth

Sermorelin
GHRH analog that stimulates your pituitary to produce its own growth hormone naturally.
Better healing during sleep & burn fat
Deeper sleep and faster recovery
Anti-aging
$200
sexual

Gooning
Triple-compound troche — Tadalafil, oxytocin & PT-141 for ultimate performance and intimacy.
Tadalafil for erections
Oxytocin for sexual desire
PT-141 for horniness
$100
energy

Vitamin B 12
Methylcobalamin the bioactive form of B12 — directly usable by the body.
Boosts energy and reduces fatigue
Nerve function & brain clarity
Promotes red blood cell production
$250
peptide
GHK-Cu
Copper peptide that signals collagen, elastin, and glycosaminoglycan production in skin.
Tighten skin and reduce wrinkles
Wound healing and hair growth
Reduce inflammation
$250
peptide
Melanotan
Alpha-MSH analog that stimulates melanocytes for a deeper, longer-lasting tan.
Natural sun-kissed tan, less UV
Reduces risk of sunburn damage
May boost libido and appetite control
$200
peptide
BPC-157
Gastric pentadecapeptide that accelerates tissue repair across gut, tendon, and muscle.
Heal injuries and joints faster
Repair and protect the gut lining
Reduce inflammation systemwide
$250
Energy
NAD+ Sublingual
Under-the-tongue NAD+ bypasses digestion for rapid bloodstream absorption.
No needles, dissolves in minutes
Cellular energy on demand
Detox
$180
Cellular energy

NAD+
Essential coenzyme that fuels cellular energy production and DNA repair in every cell.
Recharge your cells from within
Sharper focus, sustained energy
Slow aging at the cellular level
$250
Focus

Methylene Blue
Crosses blood-brain barrier & enhances mitochondrial function in brain cells.
Improves memory & focus
Mental clarity
Reduce oxidative stress
$60
Skincare

Stella +
4-in-1 compound — estriol, GHK-Cu, niacinamide & tretinoin in one topical formula.
Reverse fine lines and wrinkles
Restore collagen and skin elasticity
Brighter, firmer, younger-looking skin
$220
QUEST DIAGNOSTICS

TRT Blood Work
Total Testosterone - gold standard methodology via Quest Diagnostics.
Quest Diagnostics
Male hormone levels
Results reviewed with medical provider
$49
sexual health
PT-141
Peptide that activates sexual desire through the central nervous system, not blood flow.
Boost horniness and sensitivity
Works for men and women
Effects last up to 72 hours
$200
Hair Growth

Minoxidil/Finasteride
Dual-action combo — finasteride blocks DHT loss while minoxidil stimulates regrowth.
Stop thinning at the root cause
Visible regrowth in 3–6 months
Two proven ingredients, one protocol
$99
Focus

Modafinil
Wakefulness-promoting agent that enhances dopamine and histamine signaling in the brain.
Razor-sharp focus for 12+ hours
No jitters, no crash
Unlock peak mental performance
$99
Detox

Glutathione
Master antioxidant — a tripeptide that neutralizes free radicals and supports liver detox.
Fights oxidative stress at the source
Brighter, clearer skin tone
Boosts immune defense and recovery
$150
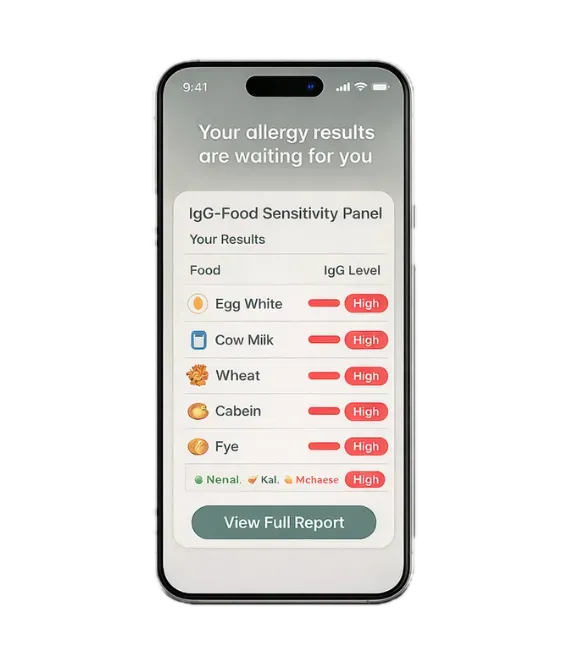
AT-HOME TEST
IgG Food Allergies
At home test kit uncovers hidden food sensitivities causing bloating and inflammation.
Finger-prick collection at home
Antibody reaction to 96 foods
Eliminate foods based on results
From $250
sexual

Tadalafil Sublingual
PDE5 inhibitor dissolved under the tongue for faster absorption than oral tablets.
Absorbs in minutes, not hours
Lasts up to 36 hours
Be ready when the moment hits
$10
SLEEP

Zolpidem
Non-benzodiazepine sedative that targets GABA receptors for rapid sleep onset.
Fall asleep in 15 minutes
Full night of restorative sleep
Wake refreshed, no grogginess
$99
AMINO ACID
Carnitine
Amino acid that shuttles fatty acids into mitochondria for energy and fat oxidation.
Turn stored fat into fuel
Enhance endurance and recovery
Upregulate androgen receptor density
$150
testosterone boost
Enclomiphene
Selective estrogen receptor modulator that signals your brain to produce more LH and FSH.
Raise testosterone without injections
Protect sperm count and fertility
Oral pill, taken once daily
$90
AT-HOME TEST

Environmental Allergy
An at-home test identifies causes of sneezing, hives, swelling, & skin rash.
Trees, Grasses, Weeds, Molds
Pet Dander, Dust Mites, Insects
88 IgE Antibody Test
$600


Answer a few medical questions and get matched with the right treatment.
No guesswork. No unnecessary options.


How It Works
Choose a treatment
Browse a curated selection of medically guided treatments designed for real needs, not trends. Each option is clearly explained so you can make an informed choice without medical jargon or confusion.
Complete a secure medical intake
Fill out a confidential health questionnaire that helps our providers understand your medical history,
goals, and current condition. Your information is encrypted and reviewed with strict privacy standards.
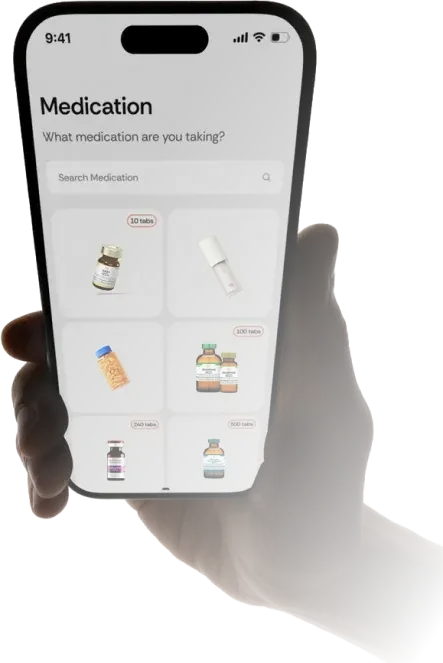
Provider review and approval
A licensed medical professional carefully reviews your intake to ensure the treatment is appropriate,
safe, and compliant. If needed, they may request additional details or lab work before approval.
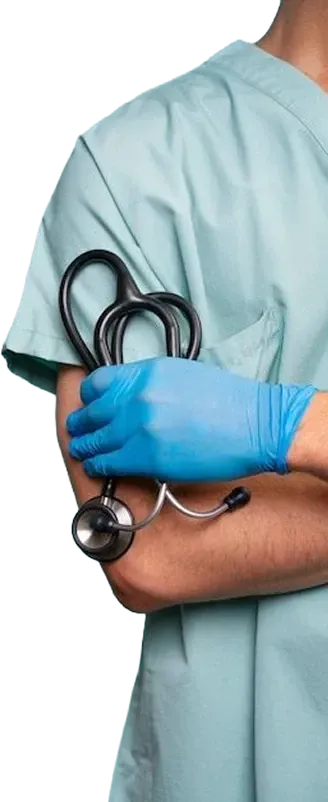

Medication delivered to your door
Once approved, your prescription is prepared by a trusted partner pharmacy and shipped directly to
you. Delivery is fast, discreet, and designed to fit seamlessly into your routine.
Private. Simple. Delivered.
Online-only medical care

Receive professional medical care entirely online, from consultation to prescription management. No waiting rooms, no appointments to commute to — everything happens on your schedule.
Discreet packaging

Your privacy comes first. All medications are shipped in plain, unbranded packaging with no indication of contents, ensuring a confidential and stress-free delivery experience.
Ongoing provider support

Care doesn’t stop once you receive your medication. Stay connected with licensed providers for follow-ups, dosage adjustments, refills, and questions through your secure patient portal.
Trust & Compliance
HIPAA-compliant platform
We use secure, HIPAA-compliant systems to protect your personal and medical data at every step —
from intake forms to ongoing care. Your information is never shared without authorization.
Licensed medical providers
All treatments are reviewed and approved by qualified, licensed medical professionals who follow
established clinical guidelines and regulatory requirements.
Partner pharmacies
Prescriptions are fulfilled by FDA licensed 503A or 503B regulated compounding pharmacies that meet strict quality, safety, and compliance standards, ensuring consistent and medical-grade formulations.
Our Experts

Dr. Gideon Kwok DO
Medical Director

Ahmed Mahdi DNP
Practitioner
Proof, Not Promises
High Standards
Higher Ratings

FAQs
Are you a pharmacy?
We are not a pharmacy. We do not manufacture, handle, or ship medications. Our partner pharmacies dispense medication after the patient has been approved for treatment and the prescription has been submitted by a licensed medical provider.
Do I need blood work?
Our medical providers recommend a baseline lab panel for a personalized treatment plan based on your biomarkers. This helps us understand your unique health profile, prescribe the right treatment at the right dose, and monitor your progress over time. We offer 16 lab panels starting at $99 through Quest Diagnostics, with results in 3-5 business days.
How does the process work?
Complete a medical intake form, consult with a licensed provider via telehealth, get your prescription sent to our partner pharmacy, and receive your medication shipped to your door.
How long does it take to get my medication?
Most patients receive their medication within 5-7 business days after provider approval.
When will I start feeling results?
Results depend on the treatment and individual health profile. Every patient's experience is different and results may vary. Your provider will discuss realistic expectations during your consultation and adjust your treatment plan based on your progress and lab work.
Who will I see for my consultation?
All consultations are conducted by licensed medical providers (DNPs or MDs or DOs) specializing in each treatment.
Do you accept insurance?
We do not accept insurance at this time. TRT can use insurance at Quest Diagnostics or Labcorp to cover the cost of blood work.
What if I'm not approved for treatment?
Our goal is to help you become healthy. Under the guidance of our medical providers, we personalize a treatment plan tailored to your goals, biomarkers, and DNA. If a specific treatment isn't right for you, a provider will recommend alternative options to help you reach your health goals.
What is your refund policy?
All prescription medications are non-refundable and sales are considered final once dispensed by our partner pharmacy. Membership subscriptions may be cancelled up to 36 hours prior to the next treatment cycle. Lab panel orders are eligible for a refund provided the order has not yet been submitted to Quest Diagnostics or Labcorp.
Our Partners







© 2026, TestosteroneShots.com PC
text (323)-283-9219
638 1/2 N Robertson Blvd, West Hollywood, CA 90069
Legal
The information and products provided on this website are for educational and wellness purposes only and are not intended to diagnose, treat, cure, or prevent any disease. All treatments require evaluation and approval by a licensed healthcare provider through a telemedicine consultation. Individual results may vary, and products carry potential risks and side effects. Certain products may not be evaluated by the U.S. Food and Drug Administration (FDA) unless explicitly stated. Services are provided by licensed physicians and nurse practitioners in states where they are authorized to practice, including California, Florida, Nevada, and other participating states. This service is not intended for medical emergencies. If you are experiencing an emergency, call 911 or seek immediate medical care. We take reasonable measures to protect personal health information in accordance with applicable privacy laws, including HIPAA. By using this website or its services, you agree to our Terms of Service, Privacy Policy, and Telehealth Consent. You must be 18 years of age or older to use this service.




